
Unprecedented overall survival in patients with 1L ESCC and PD-L1 scores ≥1%1
Key Efficacy Outcome: OS in PD-L1 ≥1%
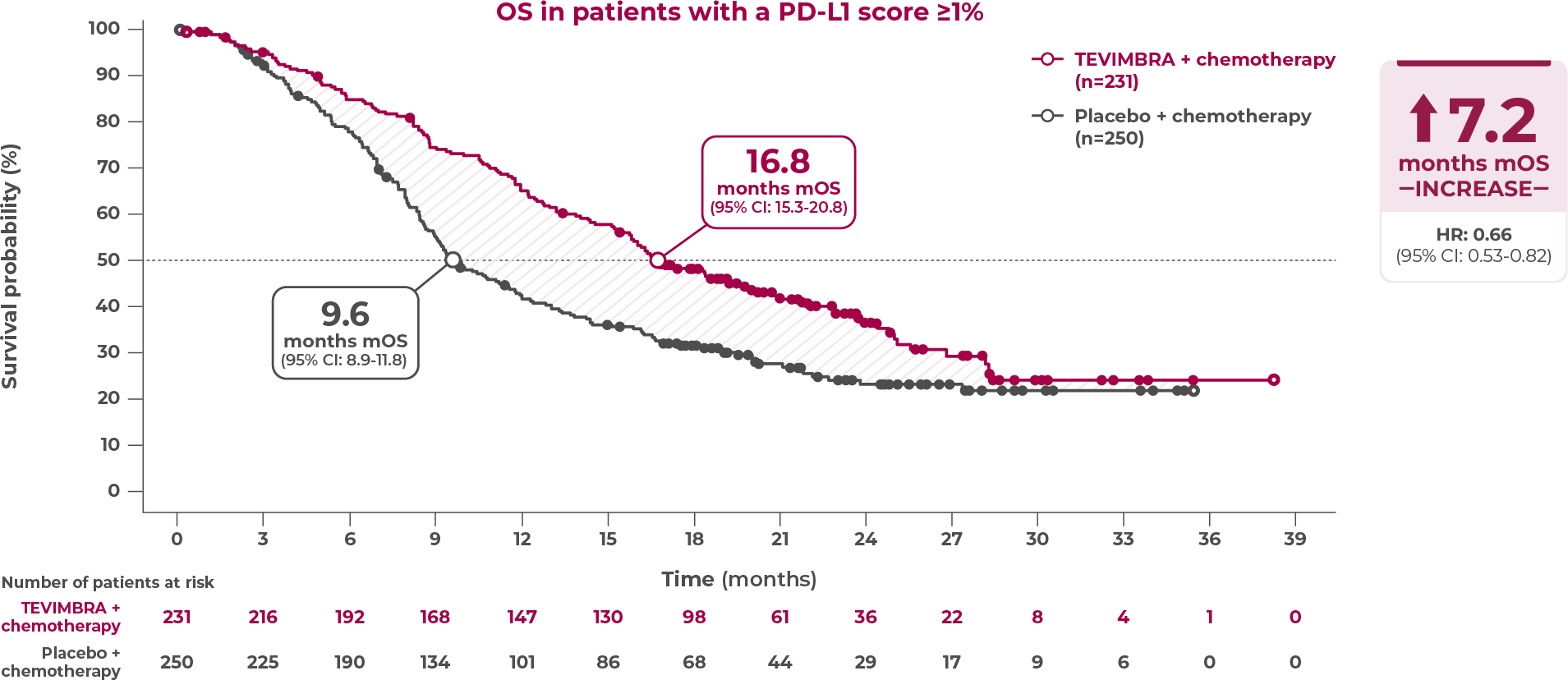
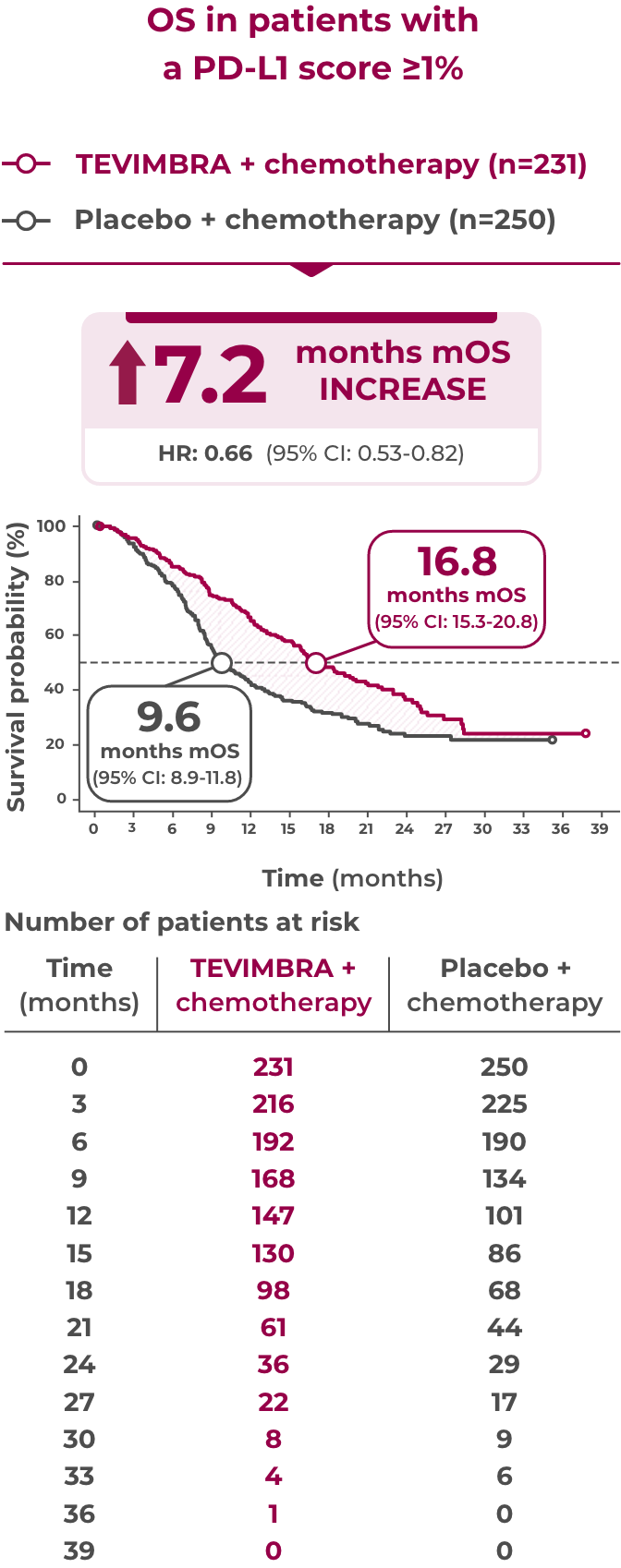
16.8 months of median overall survival with TEVIMBRA + chemotherapy vs 9.6 months with placebo + chemotherapy
Limitation: Efficacy analysis was not powered for statistical comparison and is descriptive only. No definitive conclusions can be drawn.
.
Expert Review of Overall Survival Data and NCCN Recommendations
3-Year Exploratory Follow-Up: OS in PD-L1 ≥1%
Overall survival sustained over 3 years3
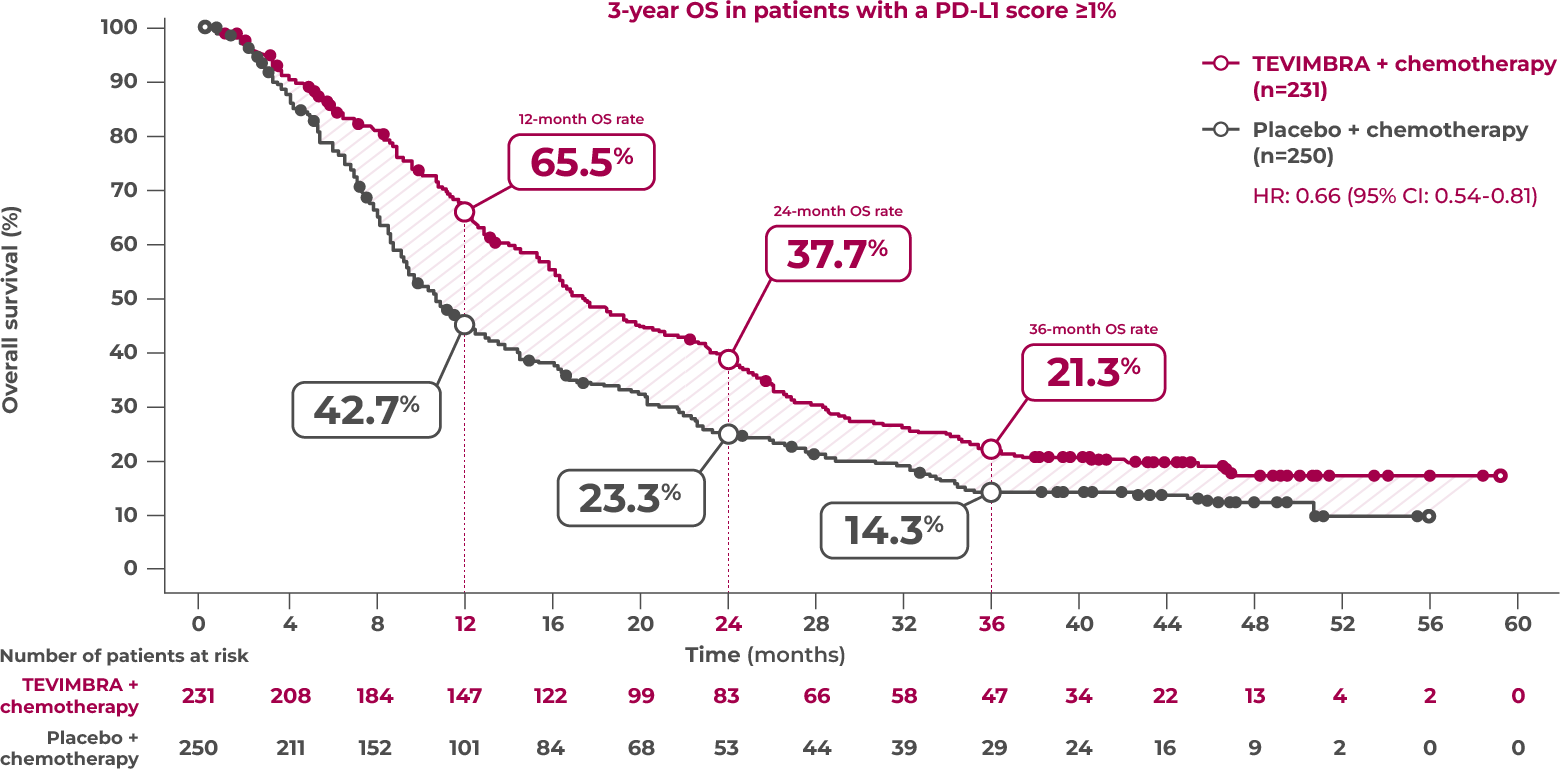
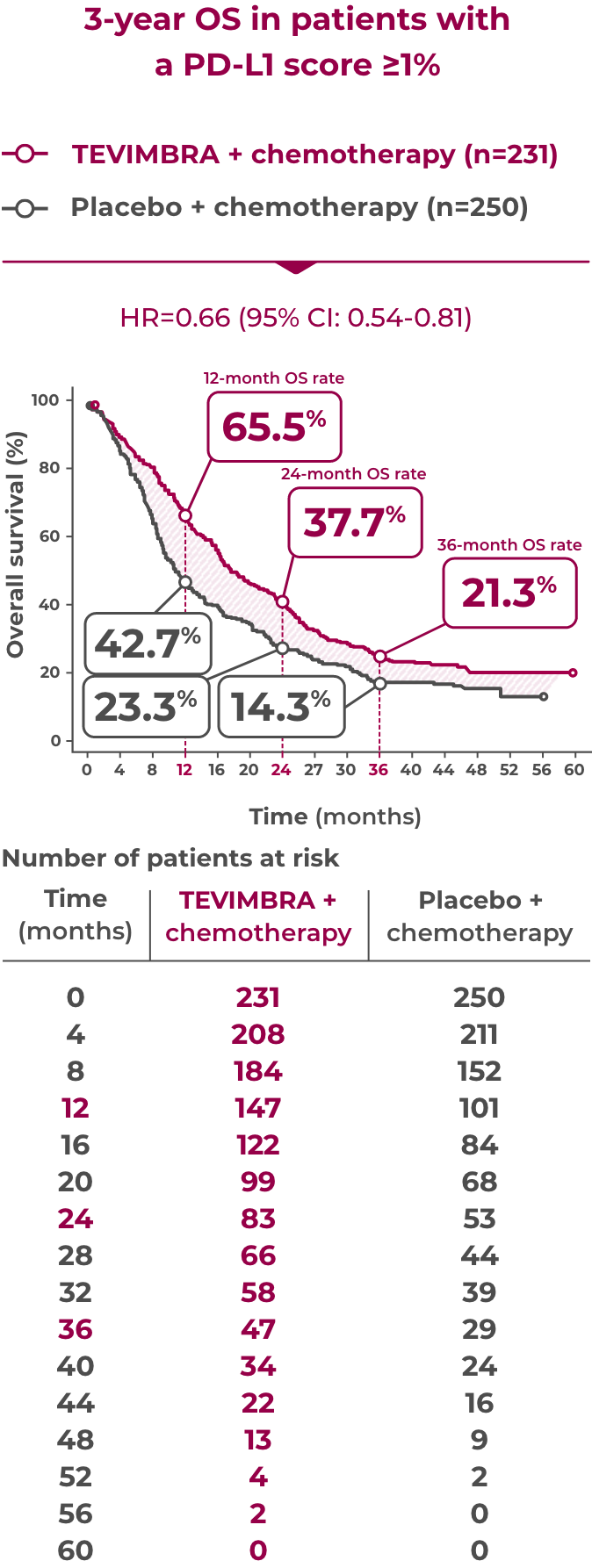
21.3% of patients were alive at 3 years with TEVIMBRA + chemotherapy (95% CI: 16.2-26.9) vs 14.3% with placebo + chemotherapy (95% CI: 10.1-19.2)
Limitation: The 3-year OS analysis was exploratory in nature and was not powered to show statistical significance. Landmark OS rates were estimated using the Kaplan-Meier method. No definitive conclusions can be drawn.
Subgroup Analyses: OS in PD-L1 ≥1%
A consistent trend in overall survival favoring TEVIMBRA + chemotherapy across multiple key subgroups3
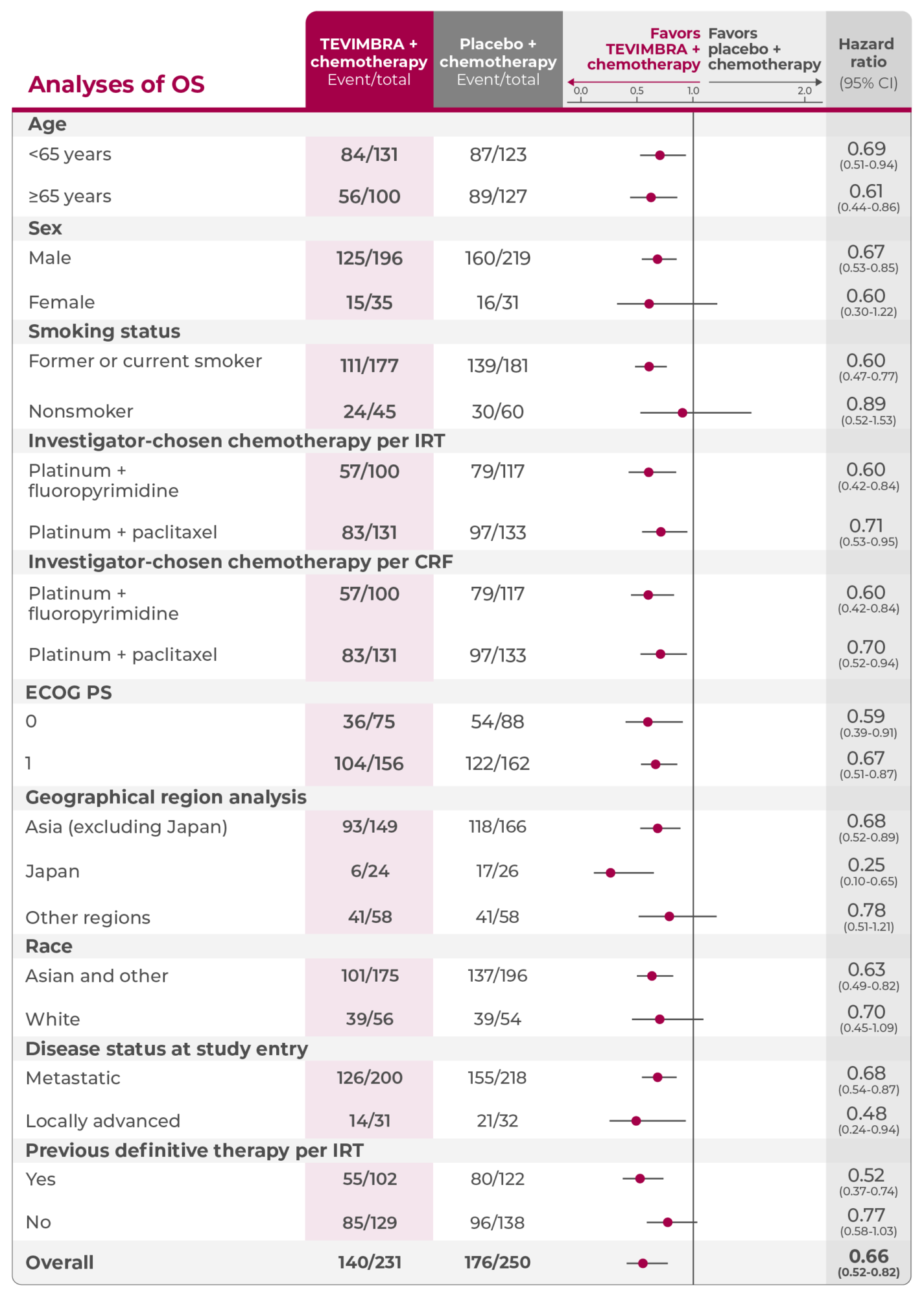
Analyses of OS
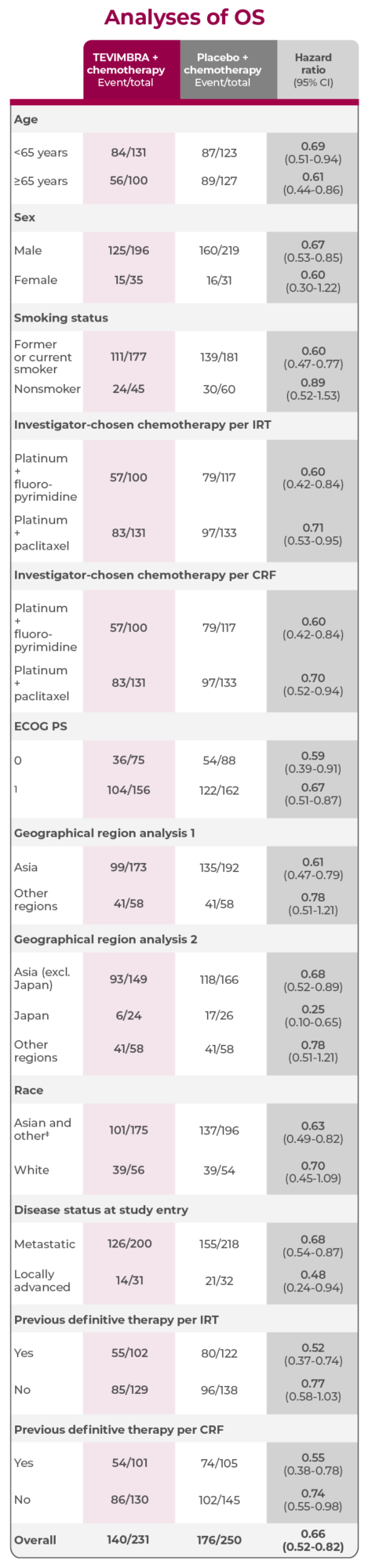
In patients with 1L, unresectable, locally advanced ESCC and PD-L1 scores ≥1% (n=63):
TEVIMBRA + chemotherapy reported 25.6 months mOS vs 11.5 months mOS with placebo + chemotherapy4
TEVIMBRA + chemotherapy reported 16.3 months mOS vs 9.4 months mOS with placebo + chemotherapy4
Frequently Asked Questions
What is the overall survival with TEVIMBRA in patients with 1L ESCC and PD-L1 scores ≥1%?
TEVIMBRA + chemotherapy delivered unprecedented overall survival results in patients with 1L ESCC and PD-L1 scores ≥1%. TEVIMBRA + chemotherapy demonstrated 16.8 months median overall survival vs 9.6 months median overall survival with placebo + chemotherapy, an increase of 7.2 months. The hazard ratio was 0.66 with a 95% confidence interval of 0.53-0.82.1
What are the 3-year results for TEVIMBRA in patients with 1L ESCC and PD-L1 scores ≥1%?
TEVIMBRA + chemotherapy provided sustained overall survival over 3 years. 21.3% of patients were still alive at 3 years with TEVIMBRA + chemotherapy vs 14.3% with placebo + chemotherapy. The hazard ratio was 0.66 with a 95% confidence interval of 0.54-0.81.3
Limitation: The 3-year OS analysis was exploratory in nature and was not powered to show statistical significance. Landmark OS rates were estimated using the Kaplan-Meier method. No definitive conclusions can be drawn.
What were the results for TEVIMBRA across key subgroups in 1L ESCC?
In a post hoc subgroup analysis, results showed a consistent trend in overall survival favoring TEVIMBRA + chemotherapy across multiple key subgroups. Notably, TEVIMBRA + chemotherapy reported 25.6 months of median overall survival in patients with 1L unresectable, locally advanced ESCC, and PD-L1 scores ≥1% vs 11.5 months median overall survival with placebo + chemotherapy.3,4
Limitation: Post-hoc subgroup analyses were not statistically powered and were descriptive only. No definitive conclusions can be drawn.
What are the NCCN recommendations for using tislelizumab-jsgr (TEVIMBRA®) in 1L ESCC?
*Category 2A Preferred if using paclitaxel and oxaliplatin or cisplatin.
†PD-L1 expression levels as determined by CPS.
‡The race subcategory “Other” included American Indian or Alaska Native, not reported, and unknown.
