
Improved ORR, mDoR, and mPFS in patients with 1L ESCC and PD-L1 scores ≥1%1
Additional Efficacy Outcomes: ORR, mDoR, and mPFS in PD-L1 ≥1%
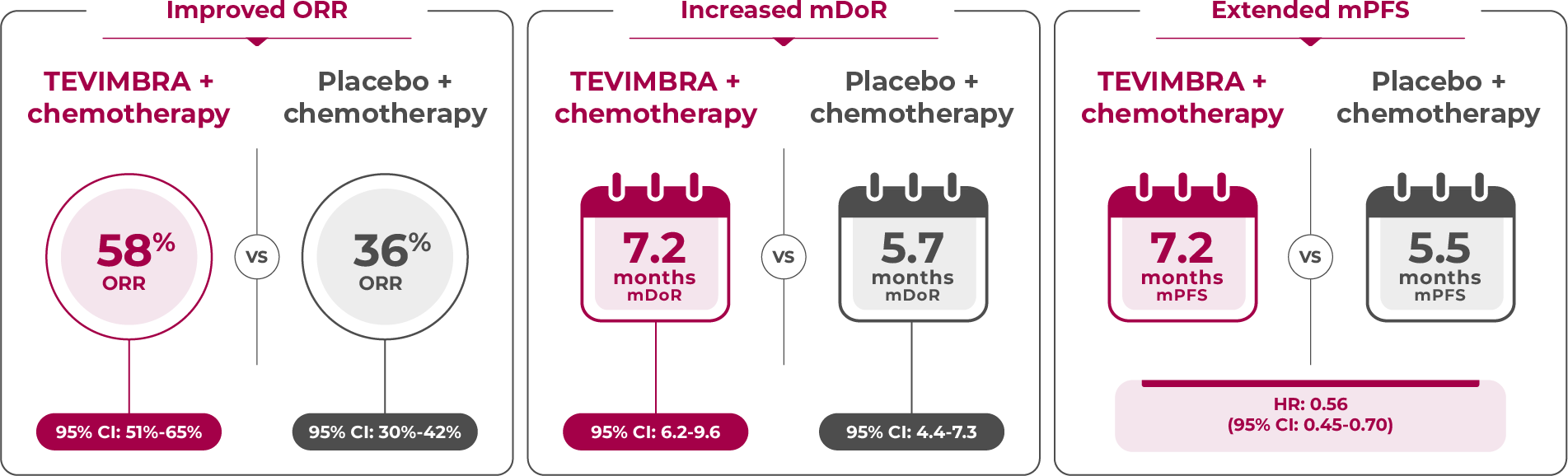
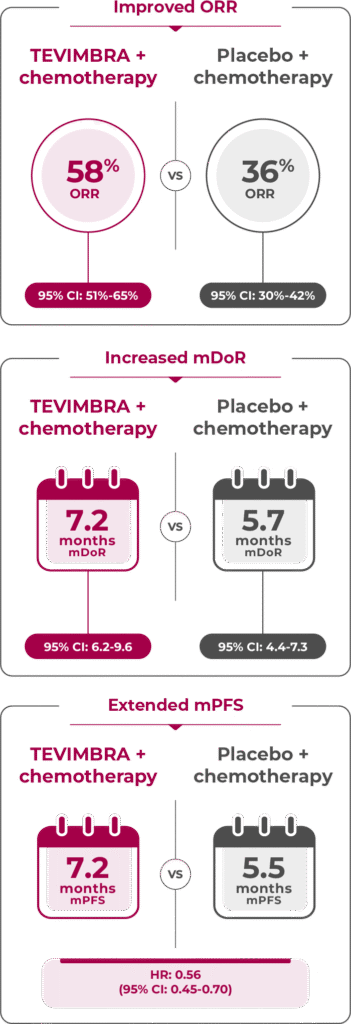
Limitation: Secondary endpoints were not powered for statistical comparison and are descriptive only. No definitive conclusions can be drawn.
Frequently Asked Questions
Expand All
What is the response rate with TEVIMBRA in patients with 1L ESCC and PD-L1 scores ≥1?
TEVIMBRA + chemotherapy demonstrated an overall response rate of 58% vs 36% with placebo + chemotherapy. Additionally, TEVIMBRA + chemotherapy extended duration of response to 7.2 months vs 5.7 months with placebo + chemotherapy. 1
Limitation: Secondary endpoints were not powered for statistical comparison and are descriptive only. No definitive conclusions can be drawn.
What is the mPFS with TEVIMBRA in patients with 1L ESCC and PD-L1 scores ≥1%?
TEVIMBRA + chemotherapy reported extended median progression-free survival of 7.2 months vs 5.5 months with placebo + chemotherapy.1
Limitation: Secondary endpoints were not powered for statistical comparison and are descriptive only. No definitive conclusions can be drawn.
1L, first line, ESCC, esophageal squamous cell carcinoma; HR, hazard ratio; mDoR, median duration of response; mPFS, median progression-free survival; ORR, overall response rate; OS, overall survival. PD-L1, programmed death ligand 1.
Reference: 1. TEVIMBRA. Prescribing Information. BeOne Medicines USA, Inc.; 2025.
